 
The first was the law of conservation of mass, formulated by Antoine Lavoisier in 1789, which states that the total mass in a chemical reaction remains constant – i.e. These experiments built upon two theories that had emerged near the end of the 18th century which dealt with chemical reactions. In the course of this research into gases, Dalton also discovered that certain gases could only be combined in certain proportions, even if two different compounds shared the same common element or group of elements. 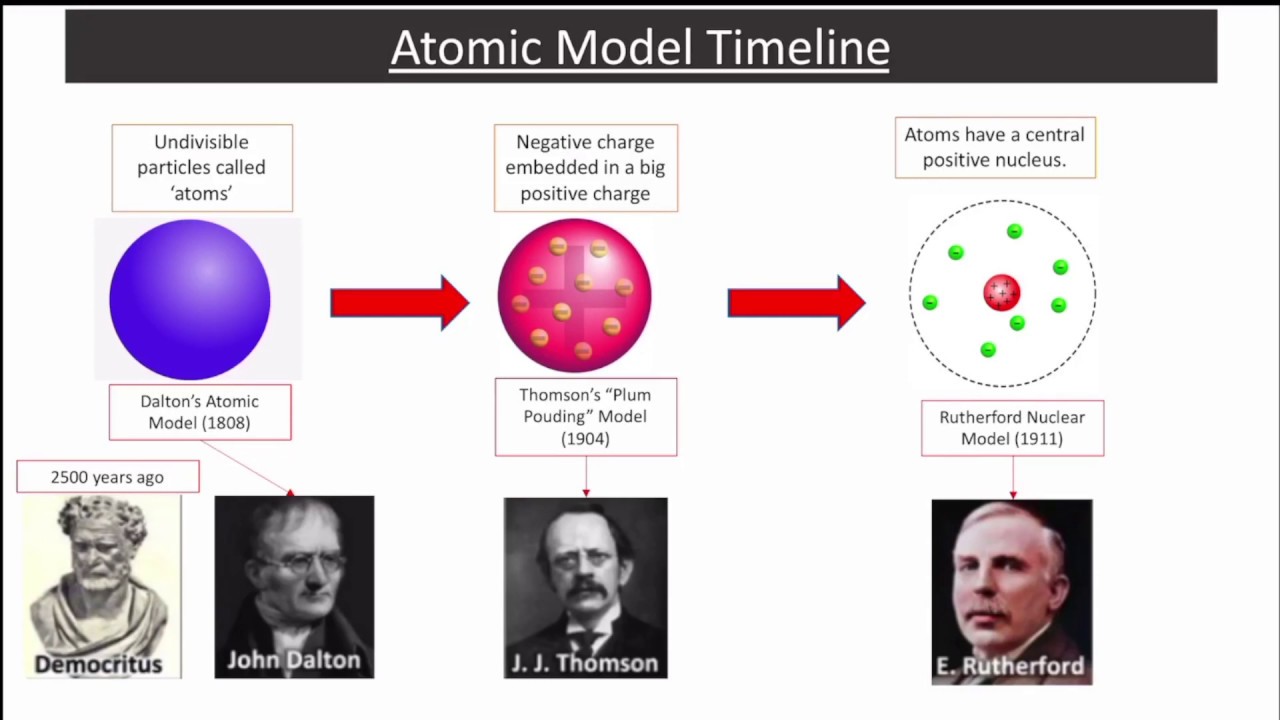
Dalton’s law of partial pressures), which stated that in a mixture of non-reacting gases, the total pressure exerted is equal to the sum of the partial pressures of the individual gases. This became the basis as Dalton’s Law (aka. noted rise in temperature using a mercury thermometer), that the corresponding expansion of air is proportionally less, the higher the temperature goes.Ī depiction of the atomic structure of the helium atom. Further, he observed that for any given expansion of mercury (i.e. He also concluded that all elastic fluids under the same pressure expand equally when heat is applied. Based on his observations of six different liquids, Dalton concluded that the variation of vapor pressure for all liquids was equivalent, for the same variation of temperature, and the same vapor of any given pressure. In his essays, Dalton described experiments in which he sought to ascertain the pressure of steam at various points between 0 and 100 ☌ (32 and 212 ☏). While there, Dalton began submitted a series of essays that outlined his experiments on the constitution of mixed gases, including the pressure of steam and other vapors at different temperatures, on evaporation. This began in 1800, when Dalton became a secretary of the Manchester Literary and Philosophical Society. 
In time, this would lead him to conclude things about how atoms interacted, the weight of atoms, and to design laws that establish atomic theory as scientific discipline.ĭalton came up with his theory of atoms as a result of his research into gases. It was at this time that John Dalton, an English chemist, meteorologist and physicist, began a series of experiments which would culminate in him proposing the theory of atomic compositions – which thereafter would be known as Dalton’s Atomic Theory – that would become one of the cornerstones of modern physics and chemistry.īeyond creating a model for atomic interactions, John Dalton is also credited with developing laws for understanding how gases work. However, it was not embraced scientifically until the 19th century, when an evidence-based approach began to reveal what the atomic model looked like. Initially, the theory appeared in thousands of years ago in Greek and Indian texts as a philosophical idea. Rutherford needed to come up with an entirely new model of the atom in order to explain his results.Atomic theory – that is, the belief that all matter is composed of tiny, indivisible elements – has very deep roots. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." 
No prior knowledge had prepared them for this discovery. Some were even redirected back toward the source. Surprisingly, while most of the alpha particles were indeed undeflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model (Quantum Model), in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. (B) According to the plum pudding model (top) all of the alpha particles should have passed through the gold foil with little or no deflection. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
